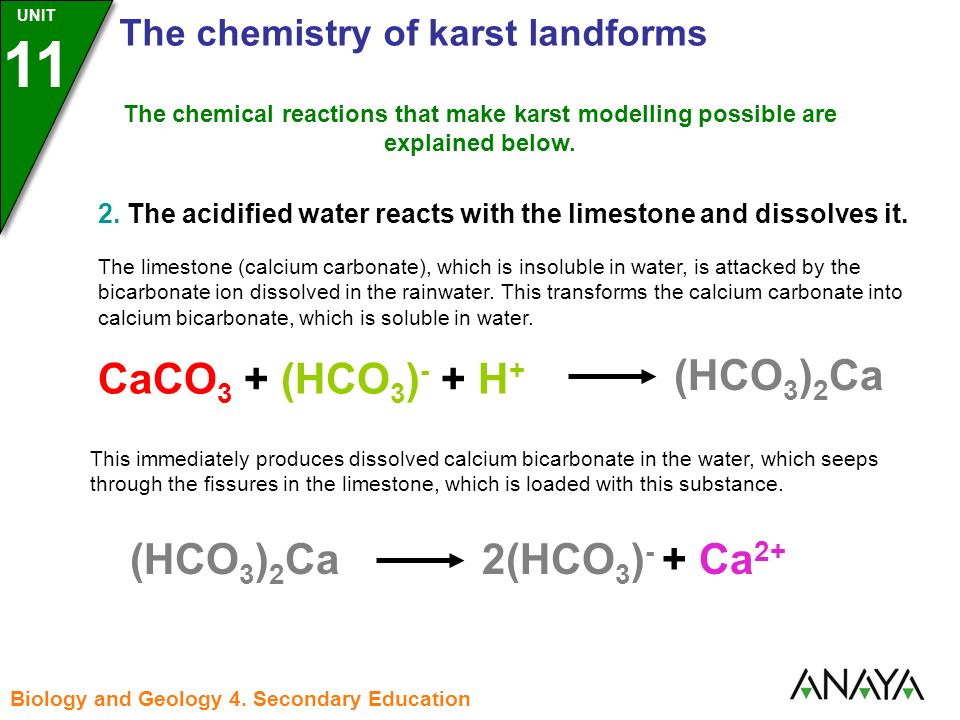
40. Consider the reaction CaCO3+2HCL (l) 》CaCl2+CO2+H2O (l).what mass of CaCO3 is required to react with 20mL 1M HCL?

The schematic diagram for the formation process of calcium carbonate... | Download Scientific Diagram
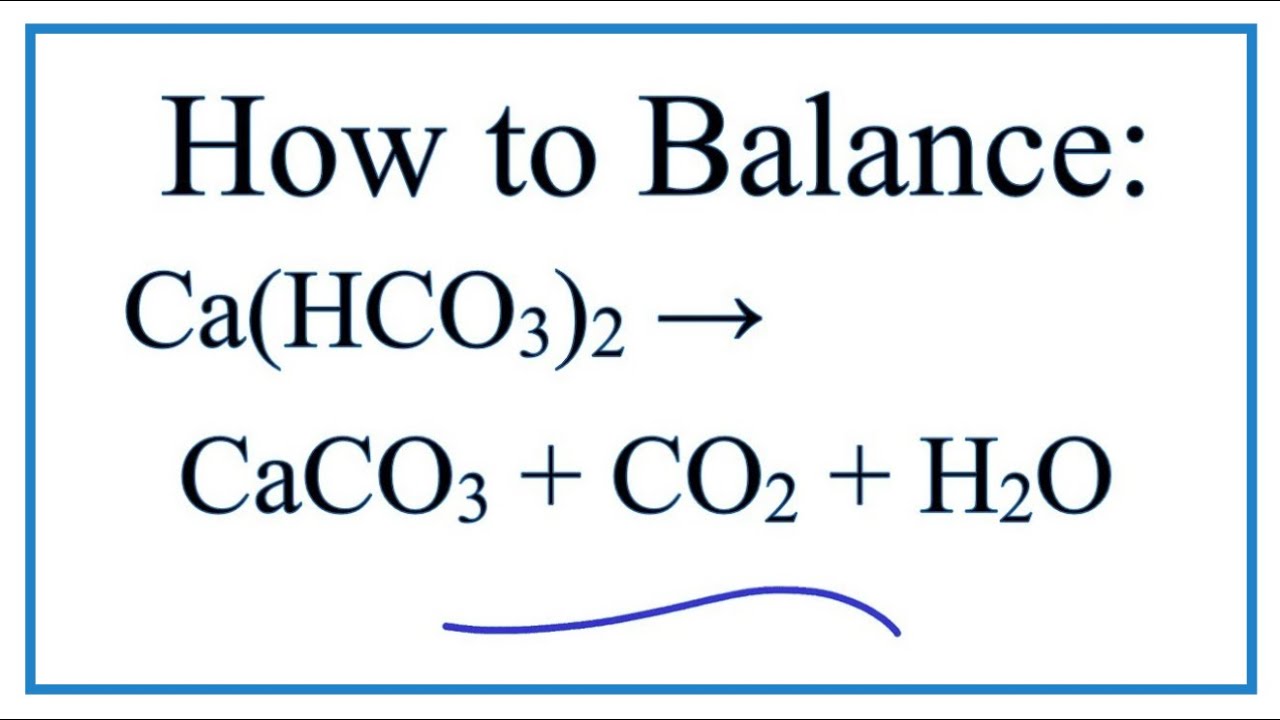
How to Balance Ca(HCO3)2 = CaCO3 + CO2 + H2O (Decomposition of Calcium hydrogen carbonate) - YouTube

Question Video: Writing a Net Ionic Equation for the Reaction of Solid Calcium Carbonate with a Hydrochloric Acid Solution | Nagwa

Ca(OH)2+CO2=CaCO3+H2O balance the equation by algebraic method or a,b,c method.ca(oh)2+co2=caco3+h2o - YouTube

SOLVED: Limestone, CaCO3, dissolves in hydrochloric acid to form calcium chloride, carbon dioxide, and water. CaCO3 + 2HCl → CaCl2 + CO2 + H2O How many grams of CaCl2 are produced from

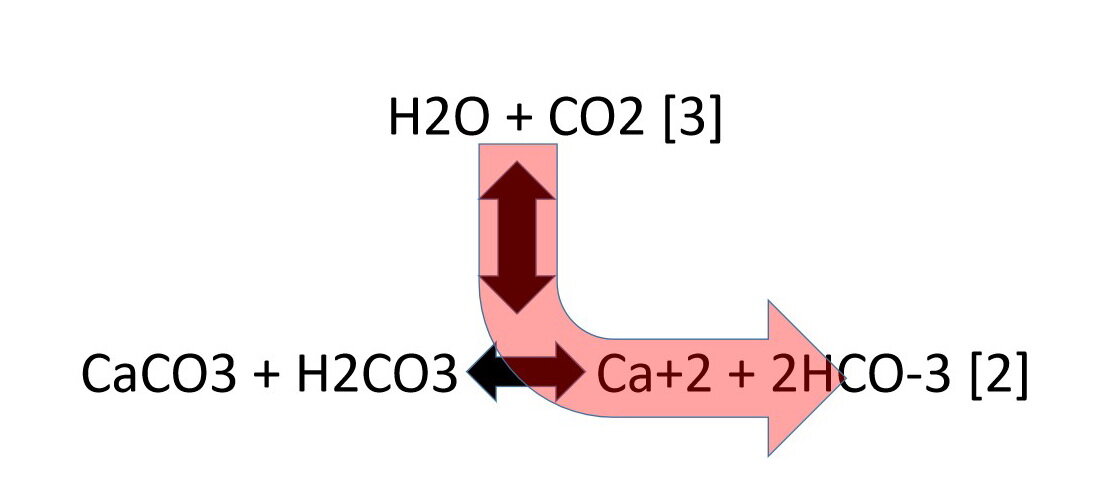
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram




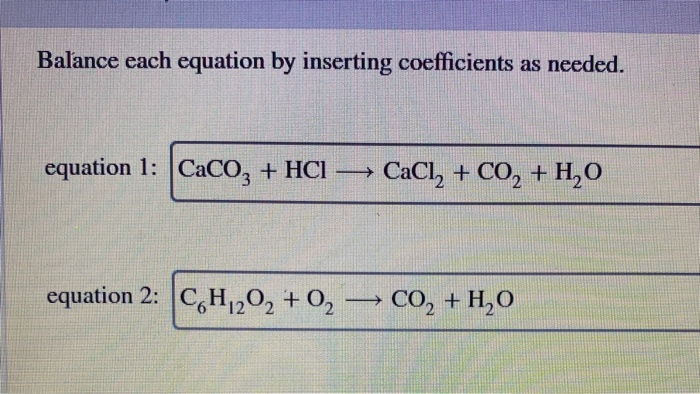



![Explain the reaction. CaCO3 + 2HCl⟶CaCl2 + H2O + CO2 [g ] Explain the reaction. CaCO3 + 2HCl⟶CaCl2 + H2O + CO2 [g ]](https://haygot.s3.amazonaws.com/questions/1352729_1287783_ans_adc66c29654147fb965de67e86f7f9a2.jpeg)