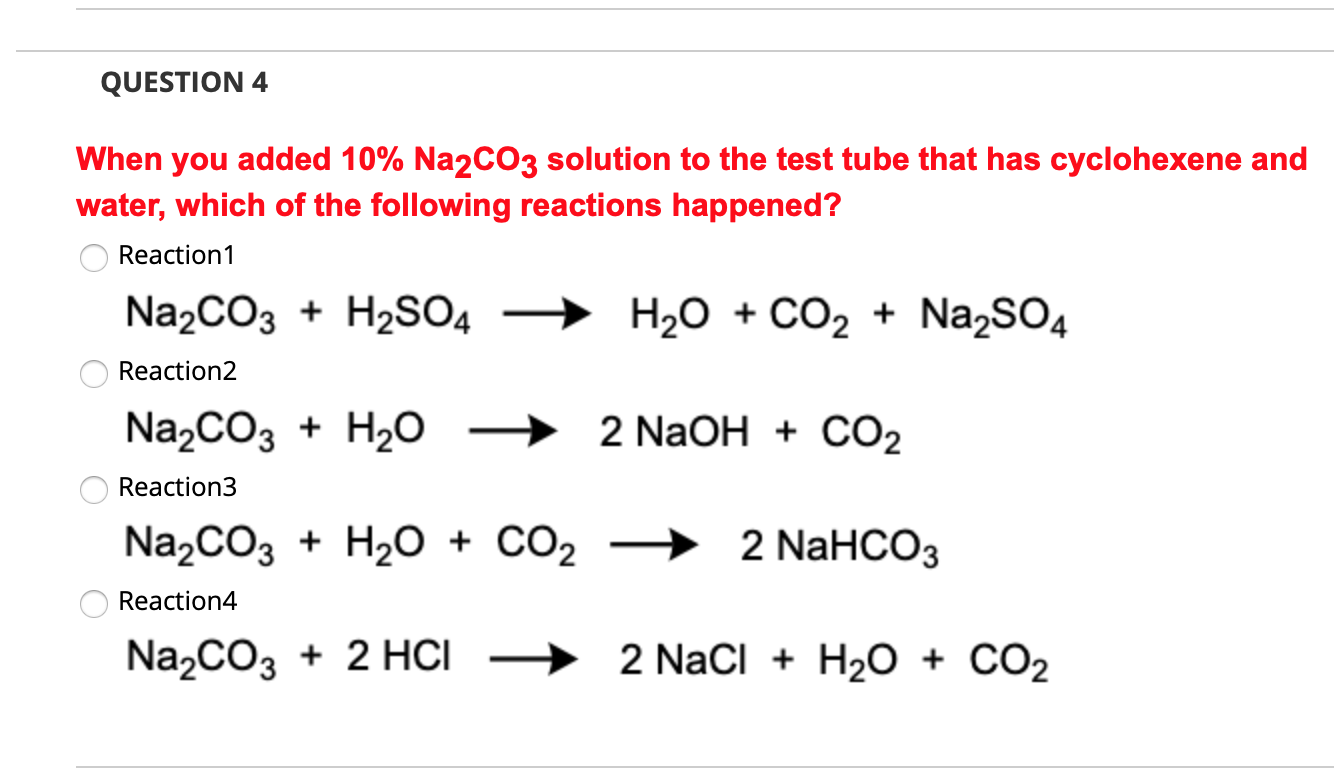
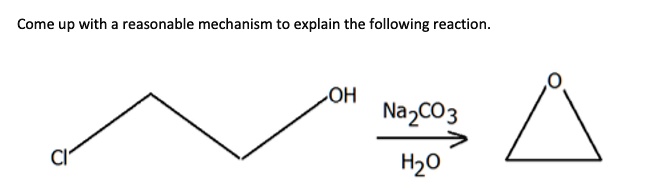
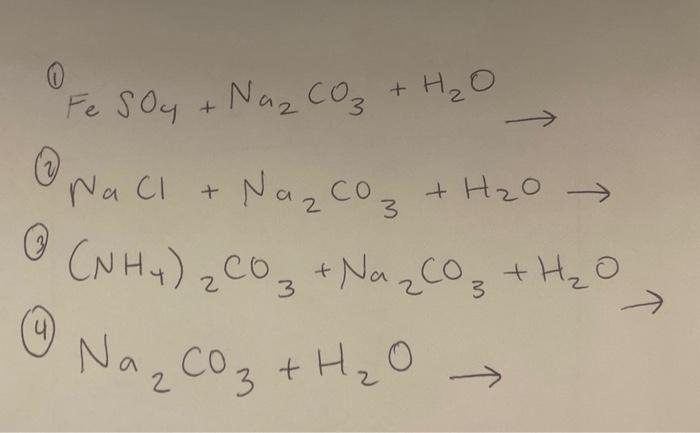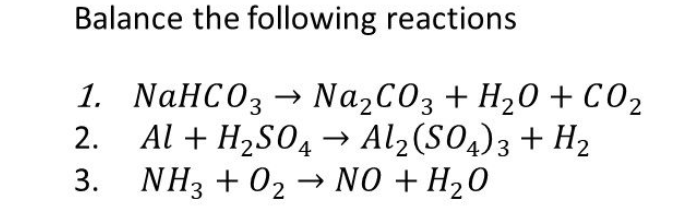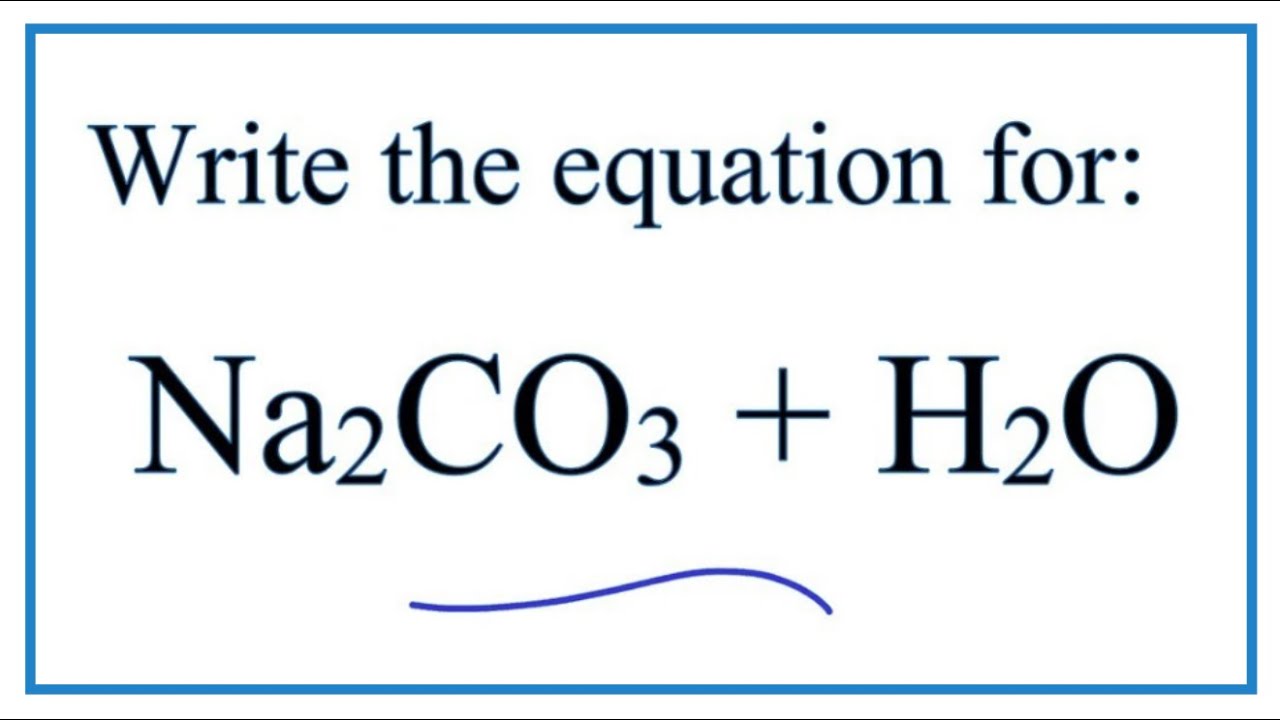How to Balance Na2CO3+H2O→NaOH+CO2 | How to Balance Na2CO3+H2O→NaOH+CO2 | By Organic Chemistry Tutorial/Inorganic Chemistry/Science - Facebook
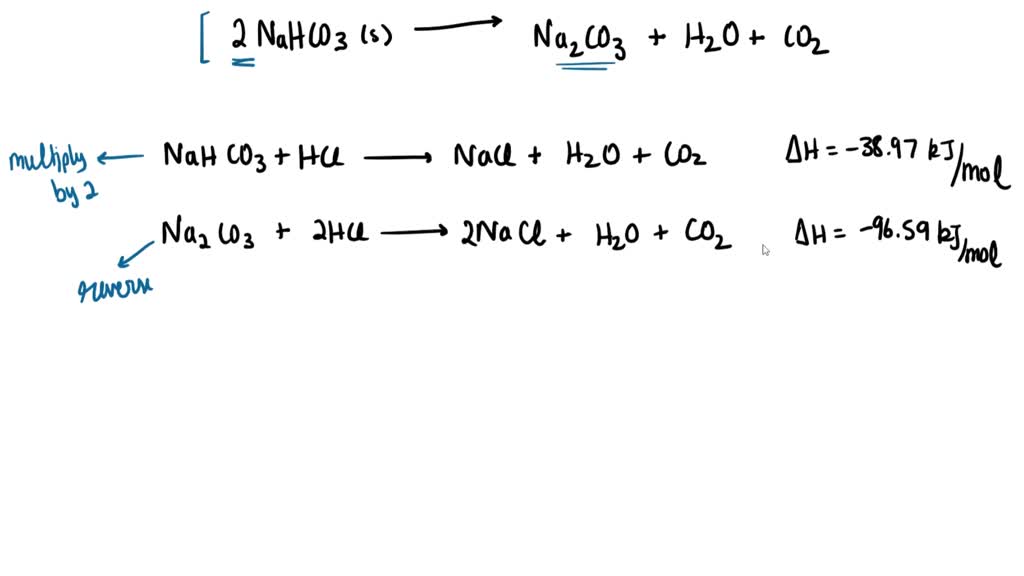
SOLVED: The following data are needed for this question: NaHCO3 (s) + HCl (aq) â†' NaCl (aq) + H2O (l) + CO2 (g) ΔH = -38.97 kJ mol-1 Na2CO3 (s) + 2HCl (